Nuclear architecture in lipodystrophic laminopathies
Mutations in the lamin A (LMNA) gene encoding lamin A/C cause laminopathies, including familial partial lipodystrophy of Dunnigan type 2 (FPLD2). FPLD2 affects adipose tissue in a depot-specific manner, leading to metabolic diseases. Some of these mutations affect the adipogenic potential of progenitor cells, as well as endothelial cell function, the latter being related to atherosclerosis, often detected in FPLD2 patients. Using patient-derived cells and engineered adipose progenitors, we investigate how lipodystrophic lamin A mutations deregulate adipose depot-specific progenitor cell nuclear architecture and function. We combine cell biology and high-throughput genomics approaches.
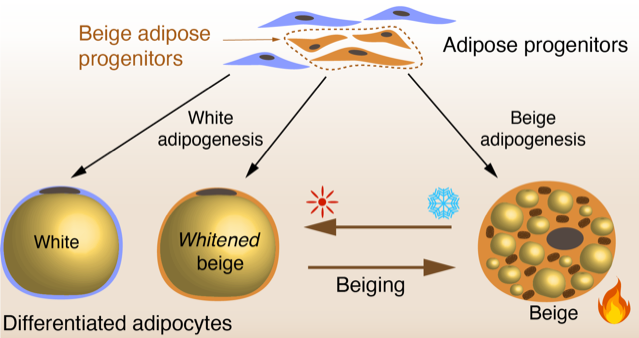
Epigenetic encoding of the thermogenic capacity of human adipose tissue
Adipose tissue has become a major research focus because of its capacity to store energy in the form of lipids in white adipocytes, and dissipate energy by heat in brown and beige – thermogenic – adipocytes. Worldwide, over 2 billion adults are overweight, including 1 billion clinically obese . The beneficial effect of thermogenic adipocytes in maintaining body weightand protecting against metabolic disorders has therefore raised strong interest in understanding their developmental origins and mechanisms of activation in response to environmental stimuli such as exposure to cold. In this project, we investigate the transcriptional and 3D epigenetic features of human beige adipocytes, that can convert between a whitened phenotype and a thermogenic beige phenotype in response to cold (β-adrenergic) stimuli (beiging). We combine cell biology, single-cell transcriptomics, epigenomics and 3D chromatin conformation studies to explore the epigenetic encoding and the 3D chromatin architecture directing this plasticity.