Background
Composed of a double lipid bilayer and a host of associated proteins, the nuclear envelope (NE) encloses the genome and physically separates the nuclear compartment from the rest of the cell. Transport across this barrier is controlled by molecular gateways called nuclear pore complexes, while underlying lamina filaments support nuclear shape and plasticity.
In addition to providing a physical barrier, the NE spatially orchestrates a range of cellular processes, mediates signal transduction, and organizes chromatin functionalization. Long considered a static entity, it is becoming clear that the nuclear envelope undergoes rapid changes in shape, integrity, and associated proteome following extracellular and intracellular cues. These changes can trigger a variety of signaling cascades that in turn affect cellular function. We are only just beginning to understand the many dynamic processes that are orchestrated by the nuclear envelope and how these affect cell fate.
About the group
The Nuclear envelope dynamics group aims to characterize pathways triggered by changes in NE topology, and to understand how such pathways affect cell fitness and fate.
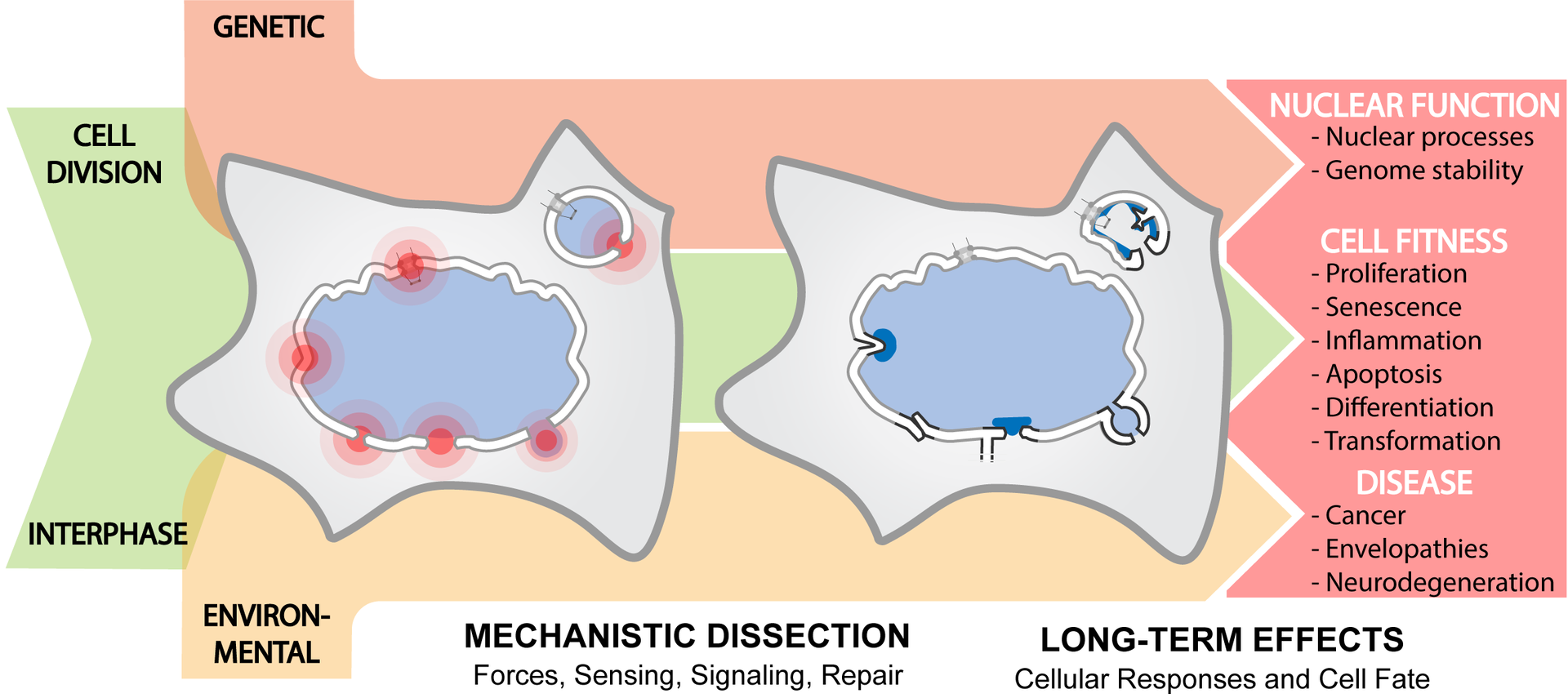
Our research focuses on
- mechanistic regulation of NE integrity during cell division, interphase, and in micronuclei.
- effects of compromised NE integrity on genome integrity, cell fitness and fate.
- consequences of genetic defects (e.g. cancer and envelopathies) on NE function and integrity.
- responses of the NE to non-genetic triggers such as mechanical stresses and their effects on cellular processes.
To address these questions, we use mechanistic cell biological studies that revolve around live-cell microscopy, proteomics, and genetics approaches in non-transformed cell lines, cancer cell lines, as well as primary cell models.
|
|
Movie 1: Cells undergo transient lapses in nuclear integrity. Nuclear ruptures are monitored by leakage of a nuclear fluorescent marker into the cytosol (in red, mCherry-NLS). These ruptures are accompanied by transient recruitment of the nuclear membrane repair machinery to the sites of rupture (in green, CHMP4B-mNG) resulting in restoration of nuclear integrity as evidenced by nuclear re-accumulation of mCherry-NLS. Movie: Coen Campsteijn/UiO. |
|
|
Movie 1: Cells undergo transient lapses in nuclear integrity. Nuclear ruptures are monitored by leakage of a nuclear fluorescent marker into the cytosol (in red, mCherry-NLS). These ruptures are accompanied by transient recruitment of the nuclear membrane repair machinery to the sites of rupture (in green, CHMP4B-mNG) resulting in restoration of nuclear integrity as evidenced by nuclear re-accumulation of mCherry-NLS. Movie: Coen Campsteijn/UiO. |
Key findings of the lab
- Unrestrained ESCRT-III drives micronuclear catastrophe and chromosome fragmentation
- The ESCRT-III machinery regulates nuclear envelope reformation during mitotic exit
Funding
- University of Oslo
- Research Council of Norway
- South-East Norway Regional Health Authorities.
Collaborators
- Winnok de Vos, Department of Veterinary Science, University of Antwerp, Belgium
- Nolwenn Briand and Philippe Collas, Department of Molecular Medicine, Institute of Basic Medicine, University of Oslo, Oslo, Norway
- Marina Vietri and Harald Stenmark, Institute for Cancer Research, The Norwegian Radium Hospital, Norway
- Sebastian Schultz and Andreas Brech, Institute for Cancer Research, The Norwegian Radium Hospital, Norway
- Roland Knorr, Max Plank Institute of Colloids and Interfaces, Department of Theory & Bio-Systems, Potsdam, Germany
- Francesca Micci, Section for Cancer Cytogenetics, Institute for Cancer Genetics and Informatics, Oslo University Hospital, Oslo, Norway
- Halim Kusumaatmaja, Department of Physics, Durham University, Durham, UK