The project is a collaboration of the Clinical Effectiveness Research Group with Bærum Hospital and research groups in Japan (Showa University) and the UK (King’s College Hospital).
Background
Removal of neoplastic, precancerous polyps in the colon during colonoscopy is the cornerstone of colorectal cancer screening.
Most of these polyps develop in the rectosigmoid colon. However, as many polyps never grow into cancer and the polyp type is difficult to determine for the endoscopist, many non-neoplastic polyps are removed today, with considerable waste of resources. A recent preliminary study indicates that a novel artificial intelligence system for computer-aided diagnosis (CAD) (EndoBRAIN®, Cybernet Systems, Corp. Tokyo) may be able to distinguish different types of polyps during colonoscopy (Mori Y, et al. Annals Intern Medicine 2018). Advanced technology in the form of Al-based pattern recognition (real-time polyp classification) can be integrated into ordinary colonoscopy practice to provide doctors with a tool to help making better and informed decision about which polyps to remove.
Objectives
The present study aims to evaluate the diagnostic performance of the EndoBRAIN system for diminutive rectosigmoid polyps during colonoscopy. To that end, we will perform a multicenter, prospective, open-label, clinical trial with 345 diminutive rectosigmoid adenomas from patients in three countries. The primary aim is to clarify whether EndoBRAIN can increase sensitivity in identification of diminutive rectosigmoid adenomas compared with visual inspection by the endoscopist alone.
Study design
This is an international, multicenter, prospective, open-label, single-arm study conducted at the Clinical Effectiveness Research Group, Vestre Viken Baerum Hospital (Norway), Showa University Northern Yokohama Hospital (Japan), and King’s College Hospital (UK). This trial is registered at the UMIN Clinical Trials Registry; registration no. UMIN00003521.
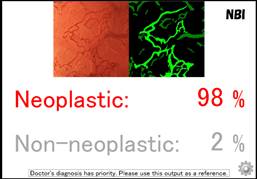
Individuals aged 18 years or older undergoing colonoscopy for any indication at the participating clinical centres, are eligible for the study. Experienced endoscopists without expertise in optical diagnosis will perform the procedures. The endoscopists assess every polyp located in the rectosigmoid colon with standard high-definition colonoscopes, first without the EndoBRAIN and then with this technology (see image above). The study flow is designed to maximize the independence between the two assessments. All polyps are then resected and subjected to histopathological assessment. Diagnostic performance with and without EndoBRAIN is compared to histopathology.
Financing
- AMED (Japan Agency for Medical Research and Development): Fees for the use of the data center and statistical analysis support will be covered by the grant.
- Olympus Inc.: Provision of endoscopic instruments for the study; research grant
- Cybernet System Corp: Provision of computer-aided diagnostic systems for the study
This is an investigator-initiated clinical trial. The companies play no role in writing the protocol, conducting the study, analyzing of the study, and reporting the study results.
Timeline
Study recruitment from April 2019 to April 2020.
Analysis of data and reporting will start after April 2020.
End date (recruitment): 10.04.2020.