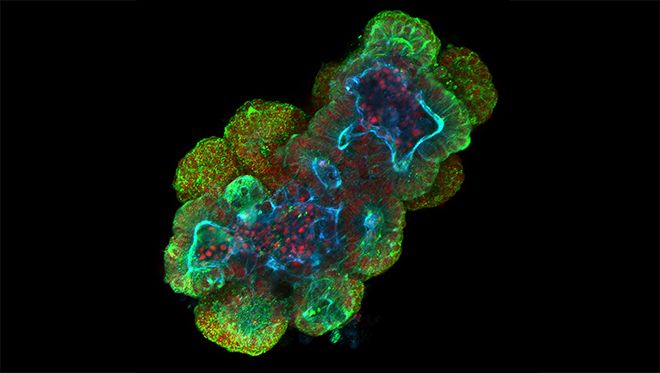
One of the most fascinating aspects of biology is developmental biology, and that means the development of an organism, from an embryo until adulthood. For some organisms, this is easily observed under the microscope, such as the elegant zebrafish embryo, which starts twitching its tail at 24 hours of age. For other organisms, development is hidden since it is within the mother, as is the case for us. ‘Why is development relevant to cancer?’ I hear you ask. Well, it turns out cancer is development gone wild. And this is why today I want to introduce you to a happy middle-ground that allows the visualisation of development of human organs. Enter the ORGANOID.
Superstar through miniature organs
The term was coined in the Netherlands about 10 years ago by Professor Hans Clevers who was already then an important researcher within the field of stem cell research1. His work on organoids has turned him into an international superstar, and I couldn’t help but feel a bit excited when I spotted him in the lobby of the Hubrecht Institute a couple of days ago. The Hubrecht Institute is in Utrecht, the Netherlands, and it is a wonderful place that focuses on developmental biology questions.
Organoids are miniature organs that recapitulate the functions of the organ they come from. They can be grown from a small amount of material, a biopsy, and can be expanded forever. The list of organoids that has been grown is long: small intestine, colon, gastric, mammary gland, pancreas, prostate, ovary, salivary gland, inner ear, eye, nasal, lung, liver… even the brain! This means that if you manage to collaborate with a clinician (and get the ethics approval), you could potentially be growing human mini-organs of the organ of your choice in no time at all. Now for those of you who don’t have access to human biopsies, you can also grow them from dissected mice.
Historic backdrop
This is of course not the first time human material is grown in the lab. Remember HeLa cells, established in the 1950s? Neither is it the first time biological material is grown in three dimensions (3D). A precursor to organoids dates back to 1906, when the hanging drop method was developed. This method, initially used to study bacteria, allows cells to grow without spreading onto a dish, as well as avoiding the drop to evaporate. I guess the revolution this time is that it is human material, that can be eternally expanded, in 3D. And the fact that they are grown in three dimensions allows the cells to organize themselves in a similar way to what they would be doing in the body, in vivo. I know what you’re thinking. What about mice? Let me tell you – a mouse is not a human.
Benefits of growing human material
Now there are different reasons for doing research. I do research because I am interested in human health. Others do research because they are interested in animal health. So depending on the biological question you are trying to address, you need to carefully consider what the appropriate model for your research is. Some diseases for example are not well modelled in mice. Obviously, experiments in humans are not possible, so growing human material, like organoids, provides huge advantages in disease modeling.
The Good
Organoids have a huge potential for applications linked to human health. They can be used for regenerative medicine, since you can expand human material from that small biopsy. And the amplification isn’t a small victory: it means you can biobank them, freeze them, thaw them, and since they can be expanded indefinitely, you won’t run out of material like you would with a regular biobanking system.
Stem cells
Why can they be expanded? The reason is stem cells. Stem cells are incredible, and given the right cocktail of growth signals, can give rise to all the cell types in a given organ. We aren’t talking of embryonic stem cells here, but rather adult stem cells, those that can be found in all of your organs. They are there so that if your organ gets damaged, the stem cells can help you out and repair the damage.
As you might have heard, our organs are not equal in this regard. Some organs regenerate much more than others, and in general it is true to say that mammals regenerate very little compared to other species. In contrast to us, zebrafish are able to regenerate most of their tissues and organs – their spinal cord, their heart, their fins, their inner ear structures, retina, kidney, liver and even the brain. Us: we can regenerate the intestine, the blood and the skin.
Having said that, the other organs still have stem cells, that can be turned on if damage occurs. So in practice it means that taking a biopsy of any organ in your body is going to contain some adult stem cells, and they are going to divide and differentiate into a miniature version of the organ. And if that doesn’t work, you can reprogram cells from a patient’s blood into stem cells, which are in this case called induced pluripotent stem cells (iPSCs), and then differentiate them into the organoid of your choice.
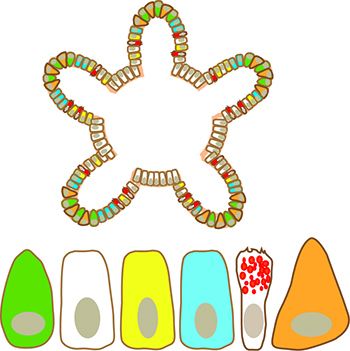 Figure (left):
Figure (left):
Colon organoids contain stem cells (green), as well as the other cell types the colon is made up of: enterocytes (white), enteroendocrine cells (yellow), trans-amplifying cells (blue), goblet cells (with red granules) and paneth cells (orange).
Illustration: Viola Lobert
Drug development
Organoids can also be used for drug development. You’ve probably heard how expensive it is to develop a new drug (about $2 billion), and this is partly due to the fact that the models used for drug development cannot predict toxicity of the drug in humans. Organoids are obviously more laborious to culture than cells grown on plastic, or feeding mice kept in captivity. But wouldn’t pharmaceutical companies ultimately save a lot of hard work and some $ if they used a more appropriate model?
Less animal testing
Another exciting implication of this technology is a reduction in animal testing. If we can use organoids to test new drugs, we won’t need mice in the same way. Cell line xenografts are currently the standard for preclinical research, and because organoids are very small (ranging from the diameter of a hair to a few millimeters for the case of brain organoids) and grow very fast, hundreds of drugs can be tested at the same time in one little plate in an incubator. Now that’s considerably more efficient than running an animal house full of thousands of mice. And that’s not even bringing in the mouse-human differences mentioned earlier.
Cancer and cystic fibrosis
Having said that, clinical applications, while seemingly limitless, have not been happening for most diseases. The problem of course is that some diseases, like cancer, are extremely complex. At the moment, there is only example of using organoids in a clinical setting: cystic fibrosis. One of the reasons it works for cystic fibrosis is that there is only one gene that is mutated here: the ion channel CFTR (in contrast, a tumour has many genetic mutations, making tumour cells more difficult to target appropriately).
Research on cystic fibrosis using organoids is done by a laboratory I am currently visiting. Jeffrey Beekman, a professor at Regenerative Medicine Center Utrecht (same building as the Hubrecht where Clevers works) has developed an assay that can predict whether a drug will work on a patient or not2. His team grows organoids from cystic fibrosis patients – patients who carry a rare CFTR mutation and where the drug of choice is unknown. They place the organoids with the drugs and use a microscope to monitor whether the organoids can swell or not. If the organoids swell, this means CFTR function is restored, and the patient will get a positive effect from using this particular drug (several approved drugs are usually tested in parallel). Then, the insurance company kicks in based on the results done in the lab, and the patient can start taking the medication. If the swelling doesn’t work, then they know there is no point using that particular drug. Wonderful, isn’t it?
So why does this give hope for cancer patients? Recently organoids have attracted a lot of attention because they predict a colorectal cancer patient’s response to chemotherapy3. This has of course wonderful consequences for the patient. Since chemotherapy works on some patients (responders) but not on others (non-responders) and has severe side-effects, it means that non-responders can avoid unnecessary treatment.
Another great piece of news: this predictive capability is not restricted to chemotherapy. One study4 directly compared patient-derived organoids grown from tumours that were treated with different anti-cancer drugs, targeting different pathways that are dysregulated in cancers, and compared how the patients responded to these exact same drugs. There was full overlap. This was for gastrointestinal cancers, so it remains to be seen if this is also the case for other cancers.
You can see why the future seems bright: a time when organoids can be grown from a patient, screened for the right drug, and the patient goes on to take that drug. This takes personalised medicine to a new level. So while there is no established pipeline using organoids for cancer therapy, it seems like we are getting close.
The Bad
Honestly I struggle to find the bad. However, I know the limitations of my model. While organoids contain all the cell types of an organ, and a similar architecture to the organ of interest, they lack blood vessels, an immune system and a nervous system. And they depend on matrigel. Matrigel is a gelatinous protein mixture that is composed of extracellular matrix components, mimicking the environment surrounding a tissue. It is made from mouse sarcoma cells, and because it is biological material, there are batch variations, which means introducing variation into your experiments.
Other limitations include size variations of organoid cultures. Those of us who have grown organoids know that some are small, some are large, even if they are neighbours in the same drop of matrigel. This may be due to differing numbers of stem cells within a single crypt, but could also be due to the matrigel, or their position within the matrigel drop, which could affect their ability to get nutrients from the overlaying medium.
Companies are developing hydrogels that are matrigel-free, and I’ve been in touch with Prellis Biologics who promise the moon. However, in the published literature, nothing so far has been better or cheaper than matrigel.
The Beautiful
They are beautiful. Because they are small, we can image them fully (even if sometimes it takes all night) and get high resolution images. I was lucky enough to get one of my pictures picked up last year by The Scientist. We would not have been able to get as high resolution images after dissecting a mouse gut, embedding it in paraffin, slicing it up and staining it.
Usually the type of technique used here is immunohistochemistry, which while informative if there are large differences in protein levels between your samples, does not give you a good overview of subcellular localisation. In our department, we use confocal microscopy routinely, and in the organoid presented here, I used super-resolution Airyscan microscopy.
Wouldn’t you agree that biology is beautiful, and would you have expected mini colons and mini small intestines to be so exquisitely organized? I am amazed every time.
Right now I am sitting in the same building as the man who started all of this. It’s raining. But I don’t care, because later today I’ll be doing more work on human organoids, in the organoid capital of the world. Here nearly all research groups work on organoids. The questions are different, but the models the same, which means that methodology moves at an incredible pace.
So now you know. Organs are no longer only growing inside an embryo. They can be grown in the lab, and you’ve learned about the good, the bad and the beautiful.
Glossary
Regenerative medicine: branch of medicine that aims to replace damaged or diseased tissues and organs to restore normal function.
Xenografts: graft/transplant from a donor of a species that is different to the recipient’s. In this case, human cells are grafted into immunocompromised mice.
Preclinical research: research to assess the safety of a drug/procedure, performed prior to starting clinical trials in humans.
Personalised medicine: medicine using the patient’s genes/proteins to prevent, diagnose or treat disease.
Paraffin: used to embed tissues, allowing them to be thinly sliced and stained for histological examination.
Want to know more?
Outreach video about my research
iBiology lectures by Hans Clevers
Notes:
1. Sato T, Vries RG, Snippert HJ, et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. May 14 2009;459(7244):262-265.
2. Dekkers JF, Wiegerinck CL, de Jonge HR, et al. A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat Med. Jul 2013;19(7):939-945.
3. Ooft SN, Weeber F, Dijkstra KK, et al. Patient-derived organoids can predict response to chemotherapy in metastatic colorectal cancer patients. Sci Transl Med. Oct 9 2019;11(513).
4. Vlachogiannis G, Hedayat S, Vatsiou A, et al. Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science. Feb 23 2018;359(6378):920-926.
Image on top, details: Small intestinal organoid (mouse) stained with anti-villin (green), phalloidin (blue) and Hoechst (red). Villin associates with the apical membrane, and phalloidin binds to the actin cytoskeleton, which also accumulates apically. Hoechst dye binds to DNA. Scale bar, 20µm.